
International University
Biotechnology
Biochemistry
Lecturer: Msc. Le Hong Phu

Topic 9: Detailed
Mechanism of Enzyme
Catalyst
•Group Members:
1. Do Ngoc Anh Huy
2. Tran Van Kha
3. Doan Luong Huy
4. Dang Duong
5. Nguyen Quang Toan
6. Nguyen Thi Huong Giang
7. Pham Vinh Phuoc
Group leader

Content:
•Definition and basic process:
1. Induced fit
2. Mechanisms of transition state stabilization
•Detailed in example to make you
understand more about mechanism of
enzyme catalyst:
Chymotripsin

Lock-key theory
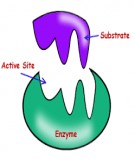
•A Specific active site for a specific substrate
How can make the
key to be fixed to the
locker???
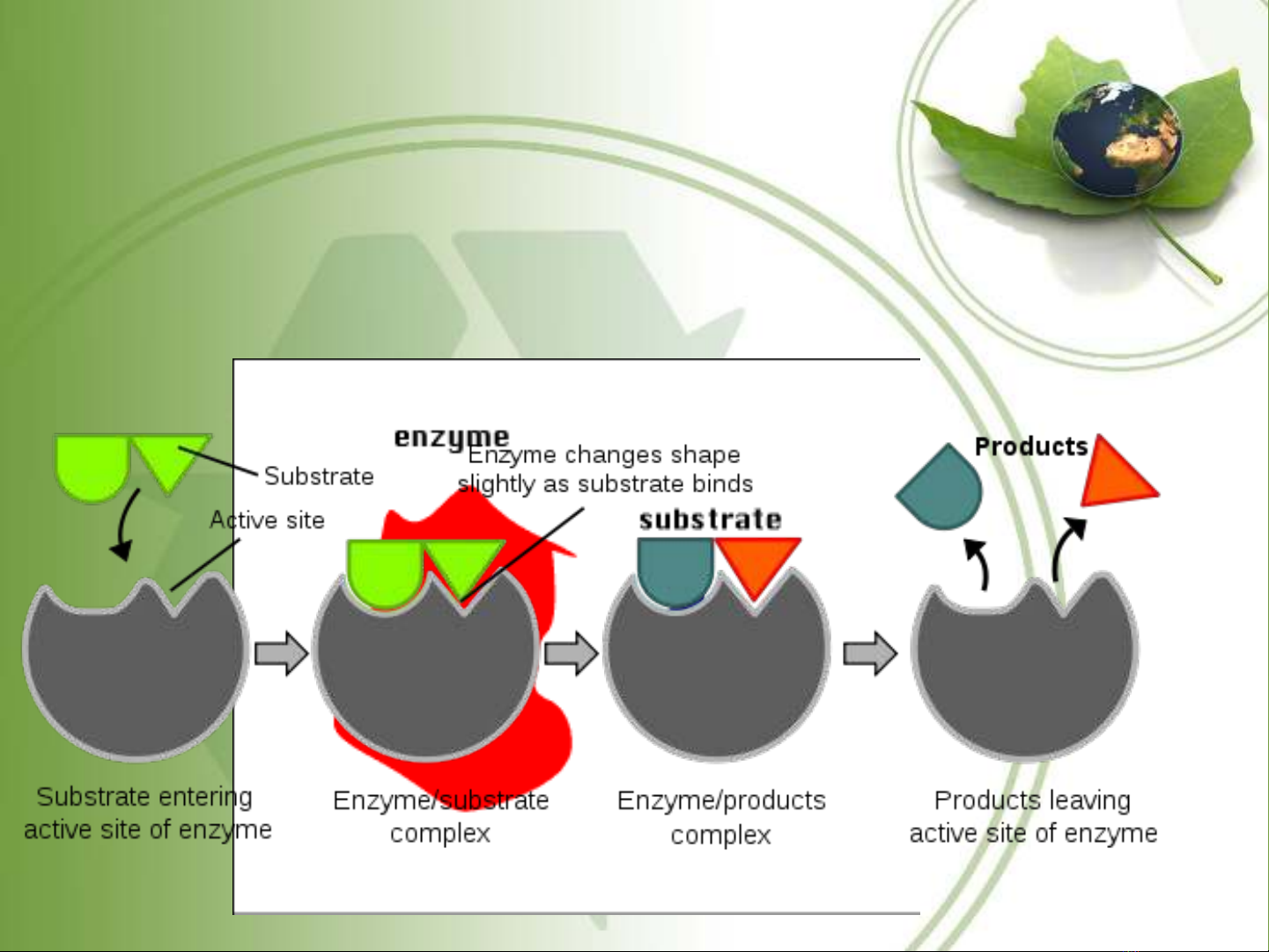
Induced fit theory
•The initial reaction between E and S is weak, but it
rapidly induce by conformational changes in the enzyme.



![Ứng dụng của enzyme: Tổng hợp, phân tích [năm]](https://cdn.tailieu.vn/images/document/thumbnail/2015/20151110/thuaco/135x160/1557937712.jpg)



![Vector chuyển gen plasmid là gì? [Hướng dẫn chi tiết]](https://cdn.tailieu.vn/images/document/thumbnail/2013/20130923/butmaulam/135x160/7661379933030.jpg)

















